Brain
Repairing the cerebral cortex: It can be done

|
A team led by Afsaneh Gaillard (Inserm Unit 1084, Experimental and Clinical Neurosciences Laboratory, University of Poitiers), in collaboration with the Institute of Interdisciplinary Research in Human and Molecular Biology (IRIBHM) in Brussels, has just taken an important step in the area of cell therapy: repairing the cerebral cortex of the adult mouse using a graft of cortical neurons derived from embryonic stem cells. These results have just been published in Neuron.
The cerebral cortex is one of the most complex structures in our brain. It is composed of about a hundred types of neurons organised into 6 layers and numerous distinct neuroanatomical and functional areas.
Brain injuries, whether caused by trauma or neurodegeneration, lead to cell death accompanied by considerable functional impairment. In order to overcome the limited ability of the neurons of the adult nervous system to regenerate spontaneously, cell replacement strategies employing embryonic tissue transplantation show attractive potential.
A major challenge in repairing the brain is obtaining cortical neurons from the appropriate layer and area in order to restore the damaged cortical pathways in a specific manner.
Train your heart to protect your mind

|
Exercising to improve our cardiovascular strength may protect us from cognitive impairment as we age, according to a new study by researchers at the University of Montreal and its affiliated Institut universitaire de gératrie de Montréal Research Centre. “Our body’s arteries stiffen with age, and the vessel hardening is believed to begin in the aorta, the main vessel coming out of the heart, before reaching the brain. Indeed, the hardening may contribute to cognitive changes that occur during a similar time frame,” explained Claudine Gauthier, first author of the study. “We found that older adults whose aortas were in a better condition and who had greater aerobic fitness performed better on a cognitive test. We therefore think that the preservation of vessel elasticity may be one of the mechanisms that enables exercise to slow cognitive aging.”
The researchers worked with 31 young people between the ages of 18 and 30 and 54 older participants aged between 55 and 75. This enabled the team to compare the older participants within their peer group and against the younger group who obviously have not begun the aging processes in question. None of the participants had physical or mental health issues that might influence the study outcome. Their fitness was tested by exhausting the participants on a workout machine and determining their maximum oxygen intake over a 30 second period. Their cognitive abilities were assessed with the Stroop task. The Stroop task is a scientifically validated test that involves asking someone to identify the ink colour of a colour word that is printed in a different colour (e.g. the word red could be printed in blue ink and the correct answer would be blue). A person who is able to correctly name the colour of the word without being distracted by the reflex to read it has greater cognitive agility.
The participants undertook three MRI scans: one to evaluate the blood flow to the brain, one to measure their brain activity as they performed the Stroop task, and one to actually look at the physical state of their aorta.
Sleep Loss Accelerates Brain Aging: Study

|
Getting a good night’s sleep may help you stay sharp mentally as you age, a new study suggests.
Researchers at Duke-NUS Graduate Medical School Singapore have found that the less seniors sleep, the faster their brains age. These findings suggest sleep deprivation is a significant factor in cognitive decline, including dementia.
Past studies have demonstrated that sleep quality and duration can affect the cognitive functions of older adults, but the latest study suggests why.
The Duke-NUS study examined tracked 66 older Chinese adults, who underwent structural MRI brain scans measuring brain volume and tests of cognitive function every two years. Researchers also asked the seniors how well - and how long - they slept each night.
To advance care for patients with brain metastases: Reject five myths

|
A blue-ribbon team of national experts on brain cancer says that professional pessimism and out-of-date “myths,” rather than current science, are guiding - and compromising - the care of patients with cancers that spread to the brain.
In a special article published in the July issue of Neurosurgery, the team, led by an NYU Langone Medical Center neurosurgeon, argues that many past, key clinical trials were designed with out-of-date assumptions and the tendency of some physicians to “lump together” brain metastases of diverse kinds of cancer, often results in less than optimal care for individual patients. Furthermore, payers question the best care when it deviates from these misconceptions, the authors conclude.
“It’s time to abandon this unjustifiable nihilism and think carefully about more individualized care,” says lead author of the article, Douglas S. Kondziolka, M.D., MSc, FRCSC, Vice Chair of Clinical Research and Director of the Gamma Knife Program in the Department of Neurosurgery at NYU Langone. The authors - who also say medical insurers help perpetuate the myths by denying coverage that deviates from them - identify five leading misconceptions that often lead to poorer care:
Study Explains How High Blood Pressure in Middle Age Affects Memory in Old Age

|
High blood pressure in middle age plays a critical role in whether blood pressure in old age may affect memory and thinking, according to a study published in the online edition of the journal Neurology.
“Our findings bring new insight into the relationship between a history of high blood pressure, blood pressure in old age, the effects of blood pressure on brain structure, and memory and thinking,” said study author Lenore J. Launer, PhD, National Institute on Aging, part of the National Institutes of Health (NIH), Bethesda, Maryland.
For the study, 4,057 older participants free of dementia had their blood pressure measured in middle-age (mean age, 50 years). In late life (mean age, 76 years) their blood pressure was re-measured and participants underwent magnetic resonance imaging (MRI) scans that looked at structure and damage to the small vessels in the brain. They also took tests that measured their memory and thinking ability.
The study found that the association of blood pressure in old age to brain measures depended on a history of blood pressure in middle age. Higher systolic and diastolic blood pressure was associated with increased risk of brain lesions and tiny brain bleeds. This was most noticeable in people without a history of high blood pressure in middle age. For example, people with no history of high blood pressure in middle age who had high diastolic blood pressure in old age were 50% more likely to have severe brain lesions than people with low diastolic blood pressure in old age.
Study Reveals Evolution at Work

|
New research by UC Santa Barbara’s Kenneth S. Kosik, Harriman Professor of Neuroscience, reveals some very unique evolutionary innovations in the primate brain.
In a study published online today in the journal Neuron, Kosik and colleagues describe the role of microRNAs - so named because they contain only 22 nucleotides - in a portion of the brain called the outer subventricular zone (OSVZ). These microRNAs belong to a special category of noncoding genes, which prevent the formation of proteins.
“It’s microRNAs that provide the wiring diagram, dictating which genes are turned on, when they’re turned on and where they’re turned on,” said Kosik, who is also the co-director of UCSB’s Neuroscience Research Institute and a professor in the Department of Molecular, Cellular and Developmental Biology. “There’s a core set with which all kinds of really complex things can be built, and these noncoding genes know how to put it together.”
Study reveals workings of working memory

|
Keep this in mind: Scientists say they’ve learned how your brain plucks information out of working memory when you decide to act.
Say you’re a busy mom trying to wrap up a work call now that you’ve arrived home. While you converse on your Bluetooth headset, one kid begs for an unspecified snack, another asks where his homework project has gone, and just then an urgent e-mail from your boss buzzes the phone in your purse. During the call’s last few minutes these urgent requests - snack, homework, boss - wait in your working memory. When you hang up, you’ll pick one and act.
When you do that, according to Brown University psychology researchers whose findings appear in the journal Neuron, you’ll employ brain circuitry that links a specific chunk of the striatum called the caudate and a chunk of the prefrontal cortex centered on the dorsal anterior premotor cortex. Selecting from working memory, it turns out, uses similar circuits to those involved in planning motion.
In lab experiments with 22 adult volunteers, the researchers used magnetic resonance imaging to track brain activity during a carefully designed working memory task. They also measured how quickly the subjects could choose from working memory - a phenomenon the scientists called “output gating.”
Family problems experienced in childhood and adolescence affect brain development

|
The study led by Dr Nicholas Walsh, lecturer in developmental psychology at the University of East Anglia, used brain imaging technology to scan teenagers aged 17-19. It found that those who experienced mild to moderate family difficulties between birth and 11 years of age had developed a smaller cerebellum, an area of the brain associated with skill learning, stress regulation and sensory-motor control. The researchers also suggest that a smaller cerebellum may be a risk indicator of psychiatric disease later in life, as it is consistently found to be smaller in virtually all psychiatric illnesses.
Previous studies have focused on the effects of severe neglect, abuse and maltreatment in childhood on brain development. However the aim of this research was to determine the impact, in currently healthy teenagers, of exposure to more common but relatively chronic forms of ‘family-focused’ problems. These could include significant arguments or tension between parents, physical or emotional abuse, lack of affection or communication between family members, and events which had a practical impact on daily family life and might have resulted in health, housing or school problems.
Dr Walsh, from UEA’s School of Psychology, said: “These findings are important because exposure to adversities in childhood and adolescence is the biggest risk factor for later psychiatric disease. Also, psychiatric illnesses are a huge public health problem and the biggest cause of disability in the world.
Researchers find retrieval practice improves memory in severe traumatic brain injury

|
Kessler Foundation researchers find retrieval practice improves memory in severe traumatic brain injury
Robust results indicate that retrieval practice would improve memory in memory-impaired persons with severe TBI in real-life settings
West Orange, NJ. January 30, 2014. Kessler Foundation researchers have shown that retrieval practice can improve memory in individuals with severe traumatic brain injury (TBI). “Retrieval Practice Improves Memory in Survivors of Severe Traumatic Brain Injury,” was published as a brief report in the current issue of Archives of Physical Medicine & Rehabilitation Volume 95, Issue 2 (390-396) February 2014. The article is authored by James Sumowski, PhD, Julia Coyne, PhD, Amanda Cohen, BA, and John DeLuca, PhD, of Kessler Foundation.
“Despite the small sample size, it was clear that retrieval practice (RP) was superior to other learning strategies in this group of memory-impaired individuals with severe TBI,” explained Dr. Sumowski.
Brain chemical ratios help predict developmental delays in preterm infants

|
Researchers have identified a potential biomarker for predicting whether a premature infant is at high risk for motor development problems, according to a study published online in the journal Radiology.
“We are living in an era in which survival of premature birth is more common,” said Giles S. Kendall, Ph.D., consultant for the neonatal intensive care unit at University College London Hospitals NHS Foundation Trust and honorary senior lecturer of neonatal neuroimaging and neuroprotection at the University College London. “However, these infants continue to be at risk for neurodevelopmental problems.”
Patients in the study included 43 infants (24 male) born at less than 32 weeks gestation and admitted to the neonatal intensive care unit (NICU) at the University College of London between 2007 and 2010. Dr. Kendall and his research team performed magnetic resonance imaging (MRI) and MR spectroscopy (MRS) exams on the infants at their approximate expected due dates (or term-equivalent age). MRS measures chemical levels in the brain.
The imaging studies were focused on the white matter of the brain, which is composed of nerve fibers that connect the functional centers of the brain.
Recurring memory traces boost long-lasting memories

|
Bonn, Germany, December 5th, 2013 - While the human brain is in a resting state, patterns of neuronal activity which are associated to specific memories may spontaneously reappear. Such recurrences contribute to memory consolidation - i.e. to the stabilization of memory contents. Scientists of the DZNE and the University of Bonn are reporting these findings in the current issue of The Journal of Neuroscience. The researchers headed by Nikolai Axmacher performed a memory test on a series of persons while monitoring their brain activity by functional magnetic resonance imaging (fMRI). The experimental setup comprised several resting states including a nap inside a neuroimaging scanner. The study indicates that resting periods can generally promote memory performance.
Depending on one’s mood and activity different regions are active in the human brain. Perceptions and thoughts also influence this condition and this results in a pattern of neuronal activity which is linked to the experienced situation. When it is recalled, similar patterns, which are slumbering in the brain, are reactivated. How this happens, is still largely unknown.
The prevalent theory of memory formation assumes that memories are stored in a gradual manner. At first, the brain stores new information only temporarily. For memories to remain in the long term, a further step is required. „We call it consolidation“, Dr. Nikolai Axmacher explains, who is a researcher at the Department of Epileptology of the University of Bonn and at the Bonn site of the DZNE. “We do not know exactly how this happens. However, studies suggest that a process we call reactivation is of importance. When this occurs, the brain replays activity patterns associated with a particular memory. In principle, this is a familiar concept. It is a fact that things that are actively repeated and practiced are better memorized. However, we assume that a reactivation of memory contents may also happen spontaneously without there being an external trigger.”
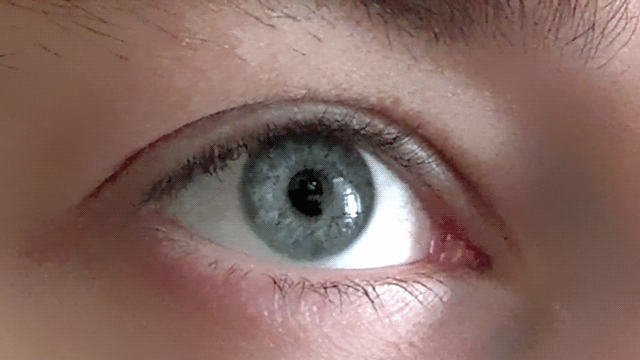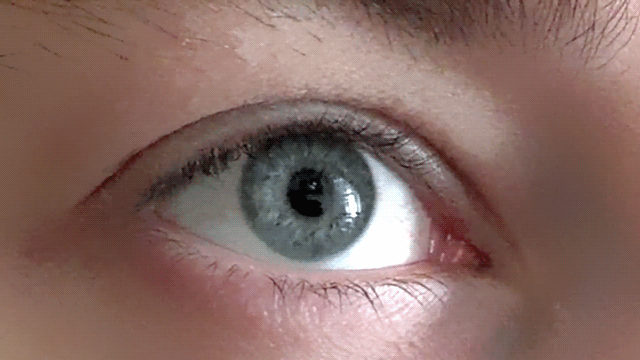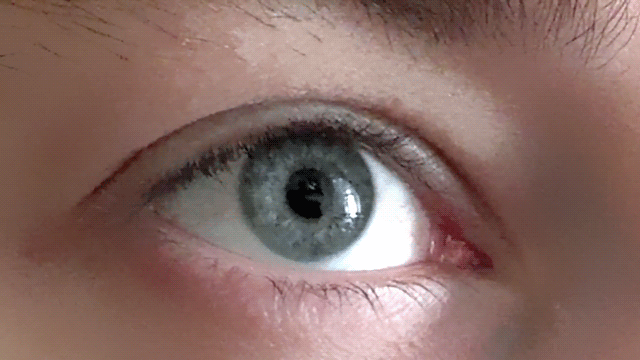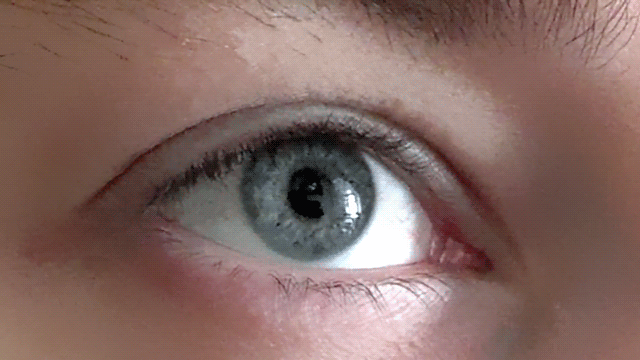
Our pupils adjust as we imagine bright and dark scenes
|
Conjuring up a visual image in the mind - like a sunny day or a night sky - has a corresponding effect on the size of our pupils, as if we were actually seeing the image, according to new research.
These findings, published in Psychological Science, a journal of the Association for Psychological Science, suggest that the size of our pupils is not simply a mechanistic response, but one that also adjusts to a subjective sense of brightness.
“Visual imagery is a private and subjective experience which is not accompanied by strongly felt or visible physiological changes,” explains psychological scientist and lead researcher Bruno Laeng of the University of Oslo. “It is a particularly difficult topic to research, as years of controversy about the nature of mental imagery testifies.”
Along with co-author Unni Sulutvedt, also from the University of Oslo, Laeng conducted a series of experiments to see whether they could tap into subjective mental imagery by monitoring pupillary size with an eye-tracking device.
Study pinpoints cell type and brain region affected by gene mutations in autism

|
A team led by UC San Francisco scientists has identified the disruption of a single type of cell - in a particular brain region and at a particular time in brain development - as a significant factor in the emergence of autism.
The finding, reported in the November 21, 2013 issue of Cell, was made with techniques developed only within the last few years, and marks a turning point in autism spectrum disorders (ASDs) research.
Large-scale gene sequencing projects are revealing hundreds of autism-associated genes, and scientists have begun to leverage new methods to decipher how mutations in these disparate genes might converge to exert their effects in the developing brain.
The new research focused on just nine genes, those most strongly associated with autism in recent sequencing studies, and investigated their effects using precise maps of gene expression during human brain development.
Italy court ruling links mobile phone use to tumor
|
|
Italy’s supreme court has upheld a ruling that said there was a link between a business executive’s brain tumor and his heavy mobile phone usage, potentially opening the door to further legal claims.
The court’s decision flies in the face of much scientific opinion, which generally says there is not enough evidence to declare a link between mobile phone use and diseases such as cancer and some experts said the Italian ruling should not be used to draw wider conclusions about the subject.
“Great caution is needed before we jump to conclusions about mobile phones and brain tumors,” said Malcolm Sperrin, director of medical physics and clinical engineering at Britain’s Royal Berkshire Hospital.
Discovery of gatekeeper nerve cells explains the effect of nicotine on learning and memory
|
|
Swedish researchers at Uppsala University have, together with Brazilian collaborators, discovered a new group of nerve cells that regulate processes of learning and memory. These cells act as gatekeepers and carry a receptor for nicotine, which can explain our ability to remember and sort information.
The discovery of the gatekeeper cells, which are part of a memory network together with several other nerve cells in the hippocampus, reveal new fundamental knowledge about learning and memory. The study is published today in Nature Neuroscience.
The hippocampus is an area of the brain that is important for consolidation of information into memories and helps us to learn new things. The newly discovered gatekeeper nerve cells, also called OLM-alpha2 cells, provide an explanation to how the flow of information is controlled in the hippocampus.
“It is known that nicotine improves cognitive processes including learning and memory, but this is the first time that an identified nerve cell population is linked to the effects of nicotine”, says Professor Klas Kullander at Scilifelab and Uppsala University.





